Sulfonyl hydrazides for Alkyl Cross-Coupling
Introduction - Major Advantages - Availability - Properties & User Guide (+ Rxn Troubleshooting)
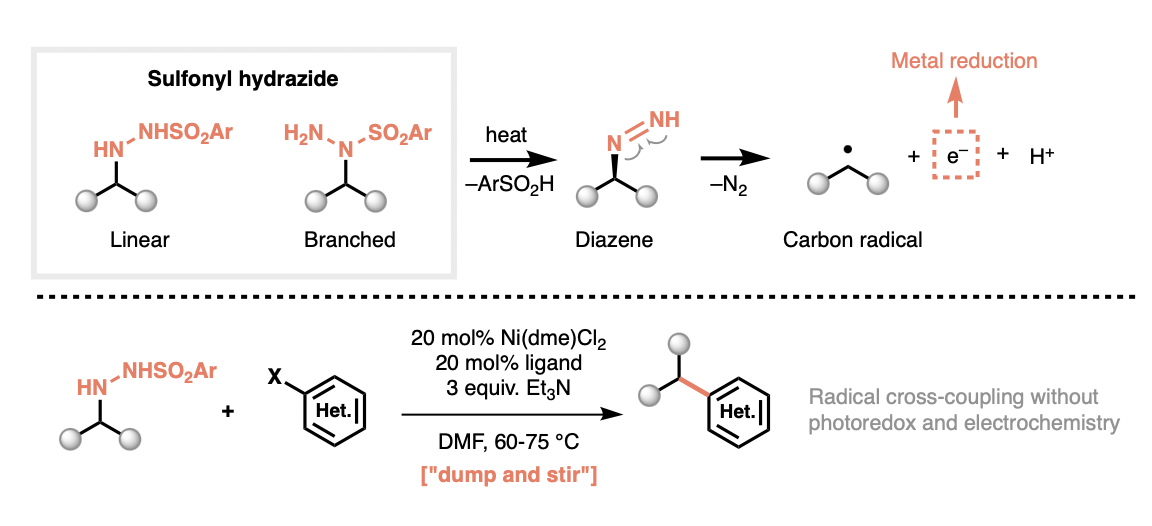
Practical building blocks for modern radical synthesis
Sulfonyl hydrazides have recently been established as highly practical radical precursors for cross-coupling, enabling the formation of C–C bonds without the need for external oxidants/reductants, photocatalysts, or electrochemical setups. In the platform reported by Baran and co-workers, both linear and branched sulfonyl hydrazides serve a dual role: they function both as the radical progenitor and as the electron donor to reductively activate the transition metal catalyst, which allows cross-couplings to proceed in a genuinely redox-neutral fashion under simple, homogeneous “dump-and-stir” conditions. This breakthrough positions hydrazide-enabled redox-neutral radical cross-coupling as a potential workhorse for alkyl coupling—analogous to Suzuki coupling for aryl coupling.
Major advantages: Facile scale-up, better yield and beyond
Conventional radical cross-couplings are powerful, but their operational complexity often hampers broad adoption. For example, photoredox-based radical cross-coupling was initially used at Bristol Myers Squibb to access an alkylated pyridine building block, and scale-up was expected to require a continuous-flow photochemical setup—an approach that is comparatively cumbersome to implement and operate. With the advent of a redox-neutral radical cross-coupling, this route was rapidly replaced by a hydrazide-based coupling, enabling straightforward scale-up without added engineering complexity.
Beyond ease of scale-up, hydrazides can also deliver improved coupling performance. As one example, installation of a hydroxyethyl group is a valuable modification because it introduces a versatile handle for downstream functionalization. Yet even this seemingly simple transformation has been hindered by the lack of a convenient hydroxyethyl radical donor: common options often fall short in reactivity, accessibility, or yield. In contrast, the hydrazide platform can leverage inexpensive 2-hydrazinoethanol as the coupling partner, and in situ activation followed by coupling provides the desired product in substantially higher yield.
Overall, hydrazide-enabled redox-neutral radical cross-coupling has the potential to transform radical coupling from an operationally demanding workflow into a simple, homogeneous “dump-and-stir” protocol, while simultaneously improving reaction outcomes. This combination of practicality and performance should make the method broadly useful for preparing advanced 3D building blocks to support the discovery and development of pharmaceuticals, spanning both small molecules and peptide-based modalities.
What is available on the market now?
Knight Chemicals is the leading company for high-quality sulfonyl hydrazides. We offer a curated, ready-to-use portfolio of hydrazide building blocks designed for redox-neutral radical cross-coupling. And because hydrazides are our core focus, we can quickly design and deliver tailor-made sulfonyl hydrazides optimized for your specific substrate, reactivity profile, and scale.
Also offered is a “plug and play” KITT 1.0 with all the reagents you need to start screening reactions. And, when you’re ready to add more substrates to your scope, expand to the Custom KITT 1.0 and choose any 12 products from the hydrazide catalogue for a flat price.
Sulfonyl Hydrazide properties & user guide
Stability and handling
Depending on the synthetic route, a sulfonyl hydrazide may be isolated either in its neutral form or as a salt. Stability can vary with structure: sulfonyl hydrazides bearing strongly electron-deficient sulfonyl groups (more electron-deficient than tosyl) may be less stable, whereas electron-deficient alkyl substituents (e.g., fluorinated alkyl groups) often enhance stability. In addition, some branched sulfonyl hydrazides can undergo sulfonyl migration to the terminal position, which is typically the thermodynamically favored isomer. All sulfonyl hydrazides supplied by Knight Chemicals are bench-stable; however, for long-term storage we recommend cold storage to best preserve purity.
General procedure
General conditions for hydrazide cross-coupling are 10–20 mol% NiCl₂•DME, 20 mol% bipyridine (ligand), and 3–6 equiv Et₃N in DMF (0.05–0.2 M) at 60–80 °C. The hydrazide/electrophile ratio can be adjusted and yields often improve when one partner is used in modest excess (1.5–2.0 equiv). All reagents can be weighed and charged under air; before heating, we recommend a brief degassing step (N₂ bubbling or freeze–pump–thaw). Reactions are typically fast and could reach near completion within a few hours. Standard aqueous workup followed by purification affords the coupling product.
Reaction troubleshooting
Problem 1: Sluggish reaction times
Most of the hydrazide remains unreacted. This usually indicates inefficient hydrazide activation. Increasing the reaction temperature often helps. Switching from Et₃N to a stronger base (e.g., tetramethylguanidine) can also improve conversion. A particularly effective strategy is to use a more electron-deficient sulfonyl group (e.g., 3,5-difluorophenylsulfonyl), which can lower the onset temperature for radical generation and significantly enhance reactivity.
Problem 2: low yields
The hydrazide is consumed, but coupling yield is low. In this case, adjusting the ligand and solvent is often beneficial. We recommend screening the following alternative ligands: 4,4'-diamino-2,2'-bipyridyl (CAS: 18511-69-8), 2-(1H-imidazol-2-yl)pyridine (CAS 18653-75-3), pyridine-2,6-dicarboximidamide dihydrochloride (CAS 1394382-56-9), and pyridine-2-carboximidamide hydrochloride (CAS 51285-26-8). Solvent effects can be substrate-dependent, but DMSO and alcoholic solvents such as EtOH or trifluoroethanol are good starting points.
More reading: Radical Cross-Coupling x AI Retrosynthesis; combining chemical and technological frontiers